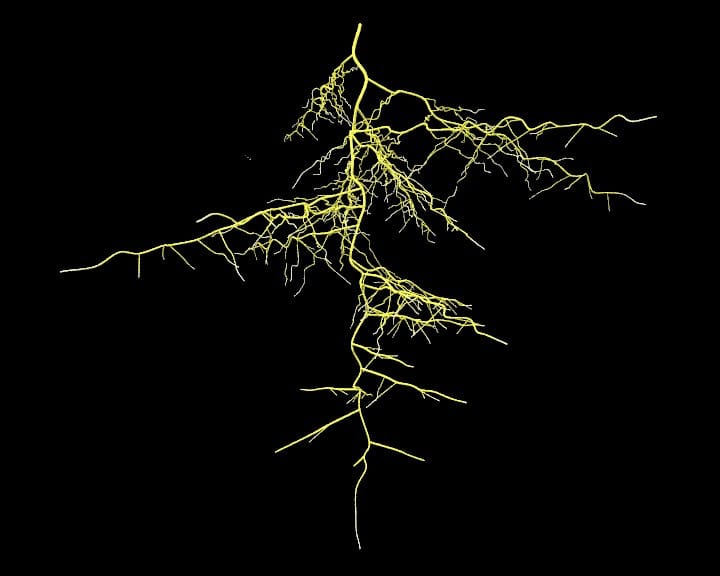
When most people (including me) hear the term ‘plant diversity’ they probably think mostly about the diversity they can see above ground. However, this is only half the story as plants have an equally diverse array of structures that grow below ground. Some below-ground traits that have so far been quantified such as root mass to root volume and fine-root diameter vary 20-fold between different plant species, highlighting how diverse even simple aspects of rooting structures can be. Numerous suggestions have been put forward as to how some of this diversity arose. One prominent hypothesis is that the climatic conditions during the Cretaceous period (approximately 145-65 MYA) promoted association with mycorrhizal fungal symbionts that drove the evolution of diverse root forms to best accommodate these associations.
An alternative hypothesis is that changes in root form were driven by historically decreasing levels of CO2, which created the need for greater gas exchange and denser leaf venation, and consequently changes in root morphology to meet water-uptake demands. However, large-scale investigations of whether these or other evolutionary pressures may underpin the vast diversity of plant root morphology are lacking. To redress this imbalance, Oscar Valverde-Barrantes and colleagues from the USA and Canada assemble datasets of numerous plant species including information on their fine-root morphology, leaf vein status and mycorrhizal fungi associations. In their study, out now in New Phytologist, they investigated the phylogenetic patterns between the traits recorded in these datasets to identify which evolutionary pressures were likely important in producing the diversity of root morphology found in plants today.
Valverde-Barrantes and colleagues find that the most substantial changes in root morphology came with the substantial shift in growth form to herbaceous growth (broadly meaning plants that do not have woody stems) in the mid-to-late Cretaceous period. They find that herbaceous plants tend to have longer and finer roots than wood-containing plants, and that this is independent of any associations with symbiotic mycorrhizal fungi. Despite substantial past discussion that mycorrhizal fungi are a significant influence on fine-root morphology evolution in plants, the authors find that this only holds true for some groups of plants and does not appear to be a broad influence in flowering plants. This suggests that root traits sometimes previously attributed to changes in associations with mycorrhizal fungi were present before such changes occurred, an idea that has gained support in some other studies recently. Moreover, associations between leaf vein density, root traits and mycorrhizal state did not hold up when phylogenetic information was incorporated.
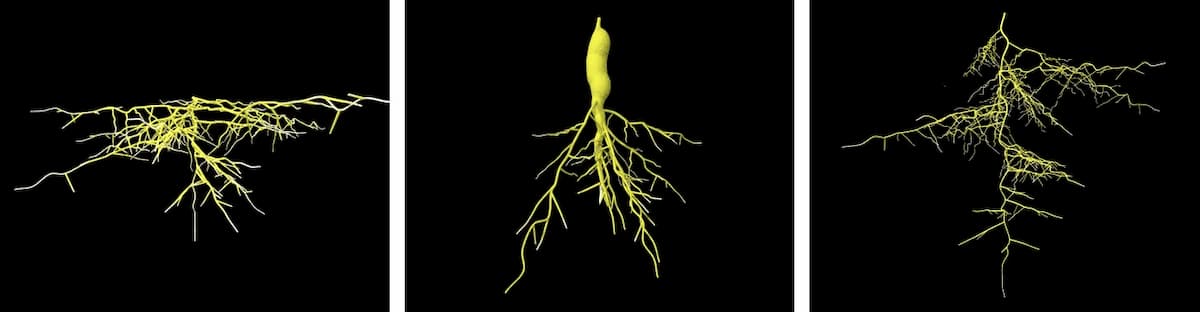
If major changes in plant root morphology of flowering plants were driven substantially by the emergence of herbaceous growth forms, this raises the question of why certain root traits co-occur with herbaceous growth. Emergence and expansion of herbaceous growth itself is believed to have been largely driven as a strategy to avoid climate extremes, which at the same time drove the evolution of faster life cycles. Root morphology traits associated with the herbaceous lifestyle include thinner roots, high root length and dense fine-roots. Such root traits are conducive to a fast-growing lifestyle with rapid nutrient acquisition but lower long-term investment in below-ground tissues, matching the faster life cycles that likely evolved to avoid climate extremes.
Valverde-Barrantes and colleagues therefore argue that the transitions to a herbaceous lifestyle was a major driver of change in root morphology in flowering plant evolution, going against other previous prominent suggestions. However, the authors do concede that there are some gaps in their analysis, particularly in surviving relatives of the earliest flowering plants. Future work will hopefully address the factors that may have driven root morphology evolution in these plants. Finally, the authors point out that their analyses may allow for accurate prediction of future changes to plant root traits in response to ongoing climate changes, and suggest that plants with thicker roots may stand to particularly benefit in the future if a warmer climate increases mineralisation. The past may therefore not only just be interesting for its own sake, but may be able to tell us something about the future!