Members of the plankton are so-called because they don’t have the ability to move against modest currents in the water bodies they inhabit, i.e. they ‘wander’ or ‘drift’ (as in the meaning of the Greek word from which they get their name). These organisms quite literally ‘go with the flow’. Plankton is broadly divided into two categories: phytoplankton (which have plant-like characteristics, including the ability to photosynthesise), and zooplankton (which don’t). Because of the photosynthetic ability of phytoplankton one might assume they play their important ecological roles in the upper-most, illuminated levels of their aquatic homes. However, this news item looks at roles of phytoplankton not only at the top of the ocean, but also in its very depths.
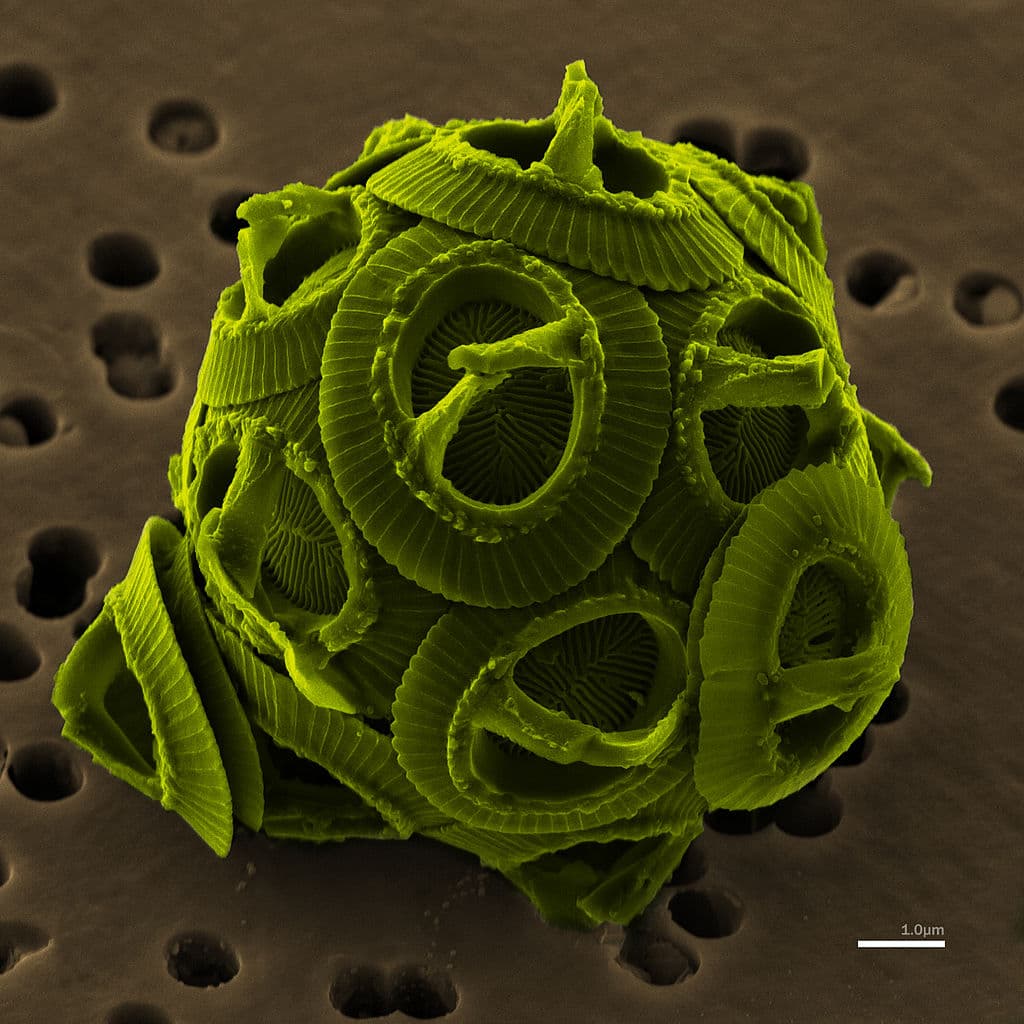
First, and demonstrating one of the many interactions and exchanges between the ocean and the atmosphere, is the finding that infection of certain phytoplankters by viruses can have an impact upon global temperatures. Miri Trainic et al. demonstrate that infection of the coccolithophore Emiliania huxleyi, by the rather unimaginatively-named E. huxleyi virus, strain 201 (EhV201), causes an increase in sea-spray aerosol (SSA) * formation. In particular, they show that parts of the external chalky coverings of the alga – the coccoliths – are shed from infected cells and released into the atmosphere above the ocean. These tiny calcareous particles are highly reflective and therefore reflect some of the sun’s radiation back into space – before it can contribute to warming of the surface of the Earth.
Somewhat balancing the global heat equation, the coccolith particles can also act as nuclei that can cause clouds to form. Some clouds help to trap radiation emitted from the surface of the Earth preventing its escape into space, thereby contributing to the greenhouse effect. Additionally, the coccolithofragments fall back down to the ocean very slowly, so persist as an effective and active component of the SSA for quite some time. As so often in nature, it’s the action of the tiny things that can have some of the biggest impacts**.
So much for living – albeit unhealthy and maybe dying – phytoplankton at the top of the ocean. Importantly, and when dead, such aquatic organisms can also play an important role in the ecology of the planet from the ocean’s depths. We are reminded of this by Sara Zaferani et al. who have examined the connection between diatoms and mercury, a toxic heavy metal. They have found that these unicellular members of the phytoplankton trap large amounts of mercury which thereby accumulates in the ‘diatom ooze’ that builds up on the seabed. Extrapolating from mercury deposits found around Antarctica, Zaferani et al. estimate that these algal remains have globally accumulated 22,000–84,000 metric tons of mercury since 1850 (when mercury pollution from human activity is judged to have begun).
Until now it had been assumed that the majority of the mercury that entered the ocean ended up in the atmosphere. Rather than any suggestion that the diatoms are killed by the mercury (although mercury can affect functioning of algae), it is suggested that the sinking of the dead diatoms ‘scavenges’ the mercury dissolved in the ocean. Arguably, this mechanism – a sort of oceanic ‘mercury pump’ – may help to clean-up upper layers of the water column removing this highly toxic heavy metal to depths where it cannot harm phytoplankton (although quite what harm it may cause to benthic biota is another matter…). Now that this discovery is published one wouldn’t be surprised to learn that entrepreneurs are already planning how to scoop up this biological bonanza, recover the mercury, and begin the whole polluting heavy metal cycling of mercury all over again.
* Typically, and as you’d probably expect, SSA contains sea salt. However, the size and shape of the coccolith fragments enable them to sink at only one-twenty-fifth of the settling rate of sea salt, thereby enriching the atmosphere in this component.
** This adds an intriguing new dimension to this organism’s relationship to the atmosphere because formerly E. huxleyi had only been known to emit DMS (dimethyl sulfoxide), a “climatically active volatile organic compound that can generate secondary aerosols through atmospheric photochemical oxidation”. Those secondary aerosols can lead to cloud formation and thereby exert an effect upon the Earth’s global heat balance…





