PAR is the acronym for Photosynthetically-Active Radiation, and refers to light with wavelengths of 400 – 700 nm, the so-called visible spectrum, from red to violet. PAR is photosynthetically-active because it encompasses the range of wavelengths absorbed by pigments involved in photosynthesis – Chlorophyll a (Chl a) and various accessory pigments, including Chlorophylls b, c, and d, and carotenoids. Although a wide range of wavelengths within that PAR band are absorbed by the multiplicity of pigments involved in photosynthesis, the energy associated with that radiation is ultimately passed to Chl a molecules that are at the reaction centres of the two photosystems (PSs) that drive photosynthesis.
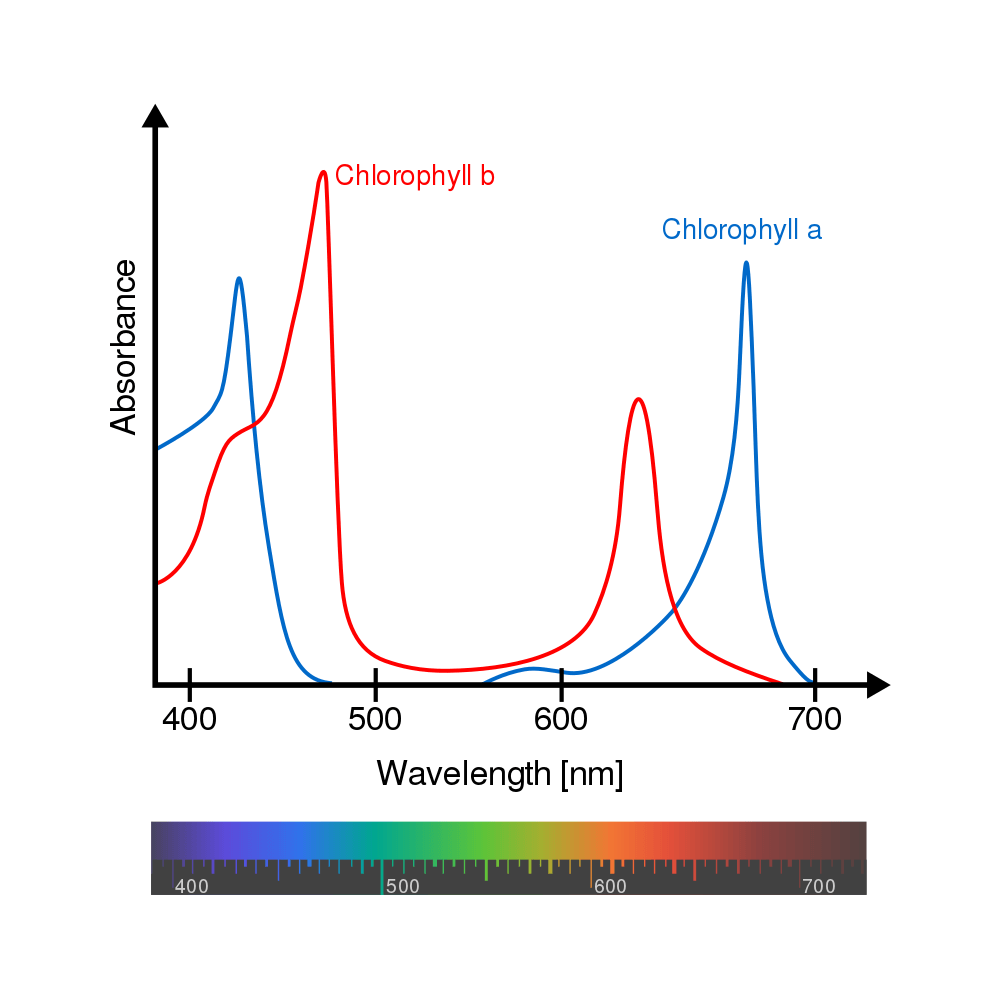
The Chl a that drives the energisation of the electrons in photosynthesis also, and importantly, absorbs light in its own right, maximally at 680 nm (in PS II) and at 700 nm in PSI, both of which are visible red wavelengths. Once absorbed by, or/and transferred to, Chl a this light energy is ultimately used to facilitate production of ATP (Adenosine TriPhosphate) and NADPH (Nicotinamide Adenine Dinucleotide Phosphate Hydrogen) in the so-called light reactions of photosynthesis (more formally, the light-dependent reactions). This ATP and NADPH subsequently participates in the light-independent reactions of photosynthesis – formerly termed the dark reactions – in which carbon dioxide is enzymically-incorporated into organic molecules for growth, etc. of the plant.
Although PAR is a term that refers to the oxygen-producing form of photosynthesis engaged in by members of the Plant Kingdom, it is also relevant to the photosynthesis undertaken by algae and seaweeds (the ‘old-fashioned’ Kingdom Prot(oct)ista), and oxygenic photosynthesis by specialised bacteria known as cyanobacteria (formerly, or still known nowadays – by those of us of a certain age and taxonomic preference – as ‘blue-green algae’) within the Domain Bacteria. And that view of photosynthesis all seemed nice and tidy and straightforward, is found in the textbooks, and is what we teach our students in the belief that this was the situation in nature.
The cosy status quo was shaken-up a little a few years ago by discovery of a new chlorophyll – Chlorophyll f (Chl f) – isolated from cyanobacteria in stromatolites in Australia by Min Chen et al. which absorbed and used light beyond the PAR range, at 706 nm. However, it had been presumed that Chl f was an accessory pigment, the ultimate driver of photosynthetic-energisation of electrons in those microbes – and as in all other oxygenic photosynthetic organisms – was Chl a. Indeed, so entrenched was this view of photosynthesis that a ‘red limit’ was believed to exist which restricted photosynthetic electron-energising to wavelengths no greater than 700 nm (which light is red). Certainly, it did not extend to use of far-red light beyond 700 nm that Chl f absorbs.
That view has now been seriously challenged by work from Dennis Nürnberg et al. using the cyanobacterium Chroococcidiopsis thermalis. When grown under 750 nm far-red illumination, photosystem II of the blue-green operated with 745 nm wavelength (solely down to Chl f), and at 727 nm for photosystem I, which may be due to Chl f (or Chl d, but – and importantly – a longer-wavelength-absorbing Chl than Chl a). Although Chl f in these complexes also acted in a light-harvesting capacity, the crucial observation is that far-red wavelengths – beyond the red limit of photosynthesis – were sufficient to drive the photochemistry of photosynthesis in these organisms. Even though Chl f can absorb at wavelengths in excess of 760 nm, the authors propose that 727 nm may be the ‘second red limit’ for photosystem II functioning. This new form of photosynthesis appears to be advantageous for organisms living in nature in deeply shaded environments where such far-red light naturally occurs. Other commentators suggest this discovery may be relevant to our searches for extra-terrestrial photosynthetic lifeforms, maybe on Mars.
Mr Cuttings’ take on this is much closer to home, and relates to recent – and on-going – discussions about where life began on Earth, and hydrothermal vents in particular. Although they are high temperature, high pressure environments devoid of surface – sun-derived – light, deep-sea hydrothermal vents may have their own in situ illumination.
That radiation, so-called geothermal radiation, is long wavelength – beyond the visible spectrum and therefore PAR – and is comprised of the electromagnetic radiation given off by hot objects in those high-temperature regions. Maybe primitive organisms evolved in such places with molecules that absorbed and exploited the long-wavelength light in an early version of photosynthesis. Some of those may have given rise to extant unicells such as Chroococcidiopsis thermalis, and others may have been the precursors of organisms that became the chloroplasts of eukaryotic cells via the operation of the Serial Endosymbiotic Theory. Subsequently, those radiation-absorbing pigments diverged and changed in response to visibly-lit environments giving rise to Chls a, b, and other accessory pigments, etc….
Thus, this exciting (yes, pun recognised and fully justified…) 21st century research may help to support the case for hydrothermal vents being the ‘cradle of life’ on Earth many billions of years ago*. In any event, textbooks will need to be amended to accommodate this new form of photosynthesis.
* Exploitation of this discovery could also help in generating plants that can use a greater proportion of the sun’s radiation than at present. For example, Rienk van Grondelle and Egbert Boeker argue that re-engineering plants to better exploit wavelengths from 700 to 900 nm is worth investigating. Or one could just try to make better use of long-wavelength-absorbing pigments apparently already present in sunflowers. Such enhanced photosynthesis and increased crop productivity might go a long way to eliminating food security issues ‘at a stroke’. Alternatively, why not engineer bacteriochlorophylls into our crop plants? These molecules (one can’t call them pigments since they absorb non-visible wavelengths), that can use irradiation up to 1050 nm wavelength, might be another as-yet-unexplored microbe-higher plant ‘symbiosis’..?







