In 2013 at the annual meeting of the Scandinavian Association of Pollination Ecologists in Finland, Dr. Amy Parachnowitsch asked the group: “Why don’t more of us measure nectar?” in her talk on floral signals, rewards and natural selection. Lively discussion during the question period ensued. The resounding conclusion was that a summary of the information available on the evolutionary ecology of nectar would be extremely useful for the community. Our recent Annals of Botany review, Evolutionary Ecology of Nectar, began with the observation that most studies on floral evolution measure selection on floral advertisements like flower size, colour, shape and even complex traits like scent but frequently ignore floral nectar*. The underlying assumption is often that these advertisements are actually indicative of a reward, and therefore are correlated with nectar traits. However, the data on nectar are scattered through the literature. So while many had impressions of the evidence available, it had not been recently or thoroughly summarized. Thus began our quest to gather and synthesize the diffuse nectar literature.
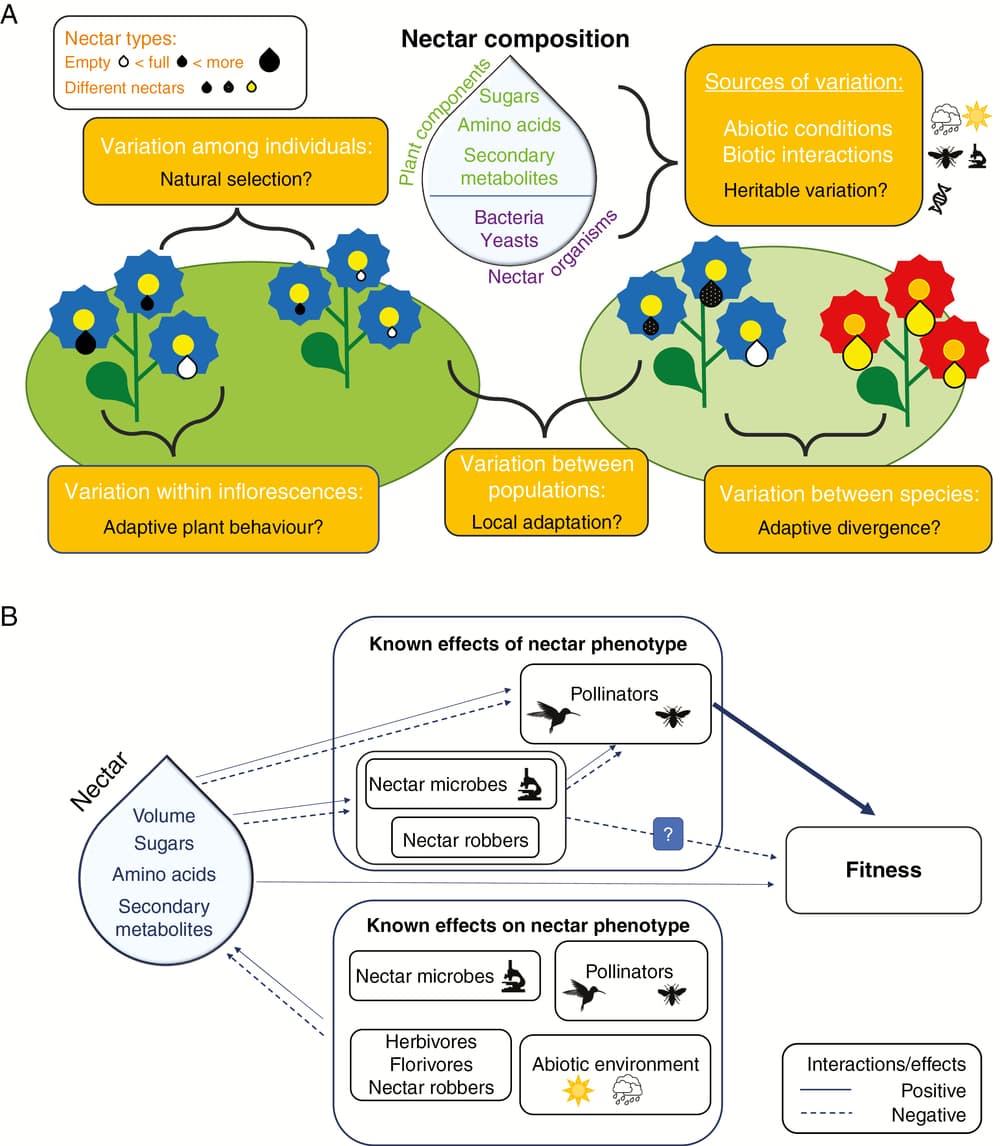
Why do we need a current summary of nectar evolutionary ecology? Flowers entice pollinators with showy advertisements like flashy colours and alluring scents. But the reward that keeps pollinators coming back, and the ultimate reason many visitors seek out flowers, is energy-rich floral nectar. Researchers hypothesise that this interaction between flowering plants and their pollinators has driven the evolution of diverse kinds of nectar across flowering plants. Evidence comes from observations such as pollination syndromes. These are suites of floral traits including nectar that are associated with specific groups of pollinators, like red trumpet-shaped flowers with lots of nectar visited by hummingbirds, and suggest that preferences by animals have been drivers of nectar evolution. However, what remains unclear is how much of nectar evolution is driven by pollinators relative to the environment (e.g. dry vs wet soils) and other visitors such as herbivores. These biotic and abiotic players can all contribute to the evolution of traits, and therefore we cannot just assume that the mutualistic interaction between pollinators and plants is the only force shaping floral traits. Furthermore, there is a lack of research on whether advertisements such as flower color, shape and scent are tied to pollinator-mediated selection on nectar. In our review, we focus on both the challenges and rewards of studying the evolution of nectar.
Nectar is a surprisingly complex trait: it is composed of sugars, amino acids, proteins and secondary metabolites that vary in identity and concentration between species, between populations and sometimes even between flowers within the same plant. Nectar volume, viscosity, pH, and added plant chemicals can all vary due to both biotic and abiotic factors. For example, visiting pollinators will not only deplete the reward available, but for some plants, also trigger how subsequent nectar is produced – and that is just one type of interaction. Calculating the heritability of nectar traits as well as selection on these traits requires careful consideration of this variation. Nectar variation may explain why many floral evolutionary ecologists have shied away from studying nectar. While there is evidence that nectar production rate and sugar concentration can be inherited and experience natural selection, the potential for the evolution of other nectar traits has received little attention.
What has been extensively explored is pollinator response to various nectar traits. Our selective literature review found that pollinators prefer nectar with amino acids but without bacteria, while responses to secondary metabolites and nectar yeasts were mixed. Further, the initial underlying assumption of advertisements being correlated to nectar within species was confirmed by our meta-analysis. Generally, nectar volume and sugar concentration are correlated with showy floral advertisements. This suggests that flowers do provide honest signals about nectar properties to potential pollinators. More importantly, these within-species correlations mean that selection can potentially act on the correlation or honesty of signals. However, there is also evidence that the strength of these correlations differ from species to species, so it may not be appropriate to generalize about honest signalling.
More recently, studies have begun to also focus on the impact of other interactions that could drive selection on nectar traits. Herbivores, nectar robbers and other animals that damage plants may indirectly influence nectar traits, including the presence of secondary metabolites in nectar. Yeasts and bacteria are ubiquitous in nectar and can alter nearly every nectar trait, which may have repercussions for pollination. Finally, environmental factors such as water and nutrient availability may constrain certain nectar traits, making a less appealing reward for floral visitors.
While everyone agreed that a review of nectar evolutionary ecology would be useful, there certainly were challenges in bringing it all together. We (the authors) might have all balked at the work to collect these nectar findings, some of it deeply buried in the literature, if we had known how much it would take at the beginning! That is why we have also made our library available for future nectar researchers to use. Our review revealed that there is substantial work to be done in order to understand nectar evolution. Our goal was to make the foundations of that research clear and easily accessible to everyone. We are excited about what future studies will reveal!
* Of which both Drs Parachnowitsch and Sletvold are guilty, and Dr Manson is only exempt because she does not measure selection in her work.




